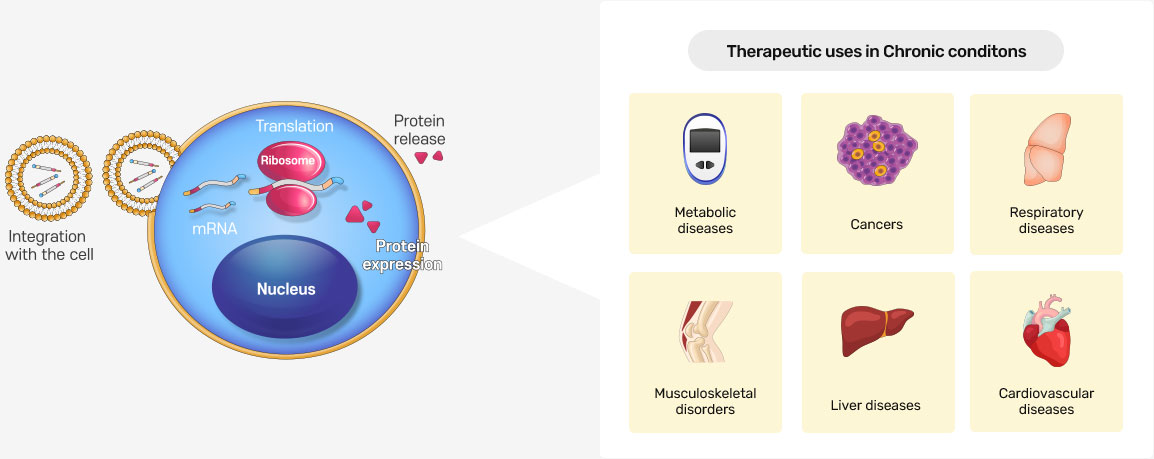
RNA Applications
Types of RNAs used as Vaccines and Therapeutics
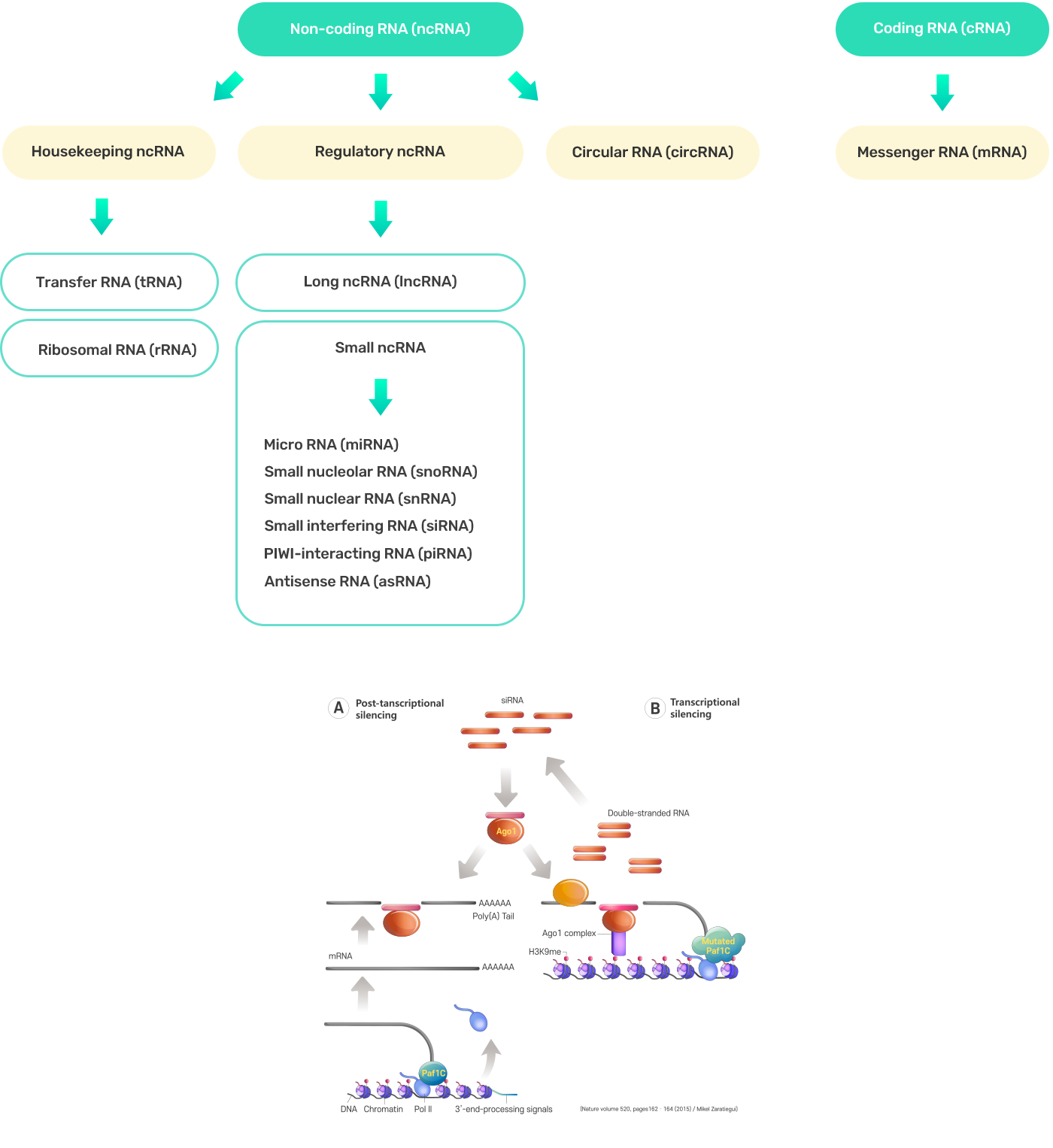
Types of RNA Applications
-
 Lipid-based Delivery
Lipid-based Delivery -
 Polymer-based Delivery
Polymer-based Delivery -
 Petide-based Delivery
Petide-based Delivery -
 Virus-like Replicon Particle
Virus-like Replicon Particle -
 Cationic Nanoemulsion
Cationic Nanoemulsion -
 Naked mRNAs
Naked mRNAs
Dendritic Cell-based mRNA vaccines
Molecular Cancer volume 20, Article number:33 (2021)
Vaccines & Therapeutics for
Infectious Diseases
Vaccines & Therapeutics for
Immuno-Oncology(Cancer)
mRNA vaccines have become a promising platform for cancer immunotherapy. During vaccination, naked or vehicle loaded mRNA vaccines efficiently express tumor antigens in antigen-presenting cells (APCs), facilitate APC activation and innate/adaptive immune stimulation. mRNA cancer vaccine precedes other conventional vaccine platforms due to high potency, safe administration, rapid development potentials, and cost-effective manufacturing. However, mRNA vaccine applications have been limited by instability, innate immunogenicity, and inefficient in vivo delivery. Appropriate mRNA structure modifications (i.e., codon optimizations, nucleotide modifications, self-amplifying mRNAs, etc.) and formulation methods (i.e., lipid nanoparticles (LNPs), polymers, peptides, etc.) have been investigated to overcome these issues. Tuning the administration routes and co-delivery of multiple mRNA vaccines with other immunotherapeutic agents (e.g., checkpoint inhibitors) have further boosted the host anti-tumor immunity and increased the likelihood of tumor cell eradication. The promising therapeutic outcomes of mRNA cancer vaccines achieved in several clinical trials against multiple aggressive solid tumors. (Molecular Cancer volume 20, Article number: 41 (2021))

LCP NP
[Molecular Therapy Vol. 26, 2018, 45:55]
-
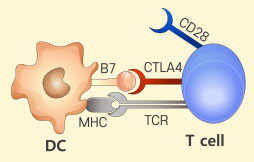 T cell anergy
T cell anergy -
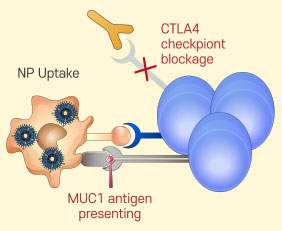 T cell activation and proliferation
T cell activation and proliferation -
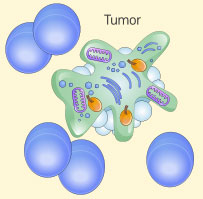 Enhanced tumor killing
Enhanced tumor killing
Vaccines & Therapeutics for
Other Chronic Diseases
Modified mRNA offers numerous advantages. One of the main advantages is that mRNA does not enter the nucleus or integrate into the host’s genome. Moreover, mRNA does not possess a risk of mutagenicity, which is the main drawback of the DNA-based delivery systems. Furthermore, modified mRNA can efficiently utilize the cell’s translation machinery to produce the therapeutic proteins of interest in a dose dependent-manner. Additionally, the safety profile of the chemically modified mRNA is better as the risk for immunogenicity remains low due to the structural and nucleotide modifications. Moreover, synthetic mRNA-based therapy is considered cheaper, faster, and easily manufactured on a large-scale. Due to the outstanding characteristics and features of modified mRNA, it has been investigated for many preventive and therapeutic pursuits in different disease states, including chronic diseases. These devastating diseases include diabetes, methylmalonic acidemia, cancer vaccination and immunotherapy, osteoporosis, osteoarthritis, osteointegration, tendinopathies, asthma, cystic fibrosis, ischemic heart disease(IHD), heart failure, myocardial infraction, atherosclerotic restenosis, chronic liver injury, progressive familial intrahepatic cholestasis type 3 (PFIC3), alpha 1-antitrypsin (AAT) deficiency, and fibrosis of the liver and lung. (Biomedicine & Pharmacotherapy, Volume 145, January 2022, 112385)
-
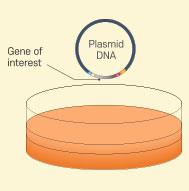 in vitro Transcription
in vitro Transcription -
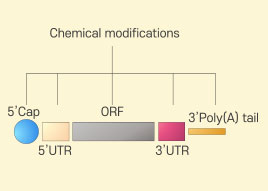 Modified mRNA
Modified mRNA -
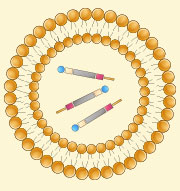 Upload into delivery system
Upload into delivery system -
 in vivo Administration
in vivo Administration
Molecular Cancer volume 20, Article number:33 (2021)